 Considering the inclusion of more topics, longer time of experimentation, size of the sample, and explicit study of their behavior, for instance, can help establish the veracity of the future results. 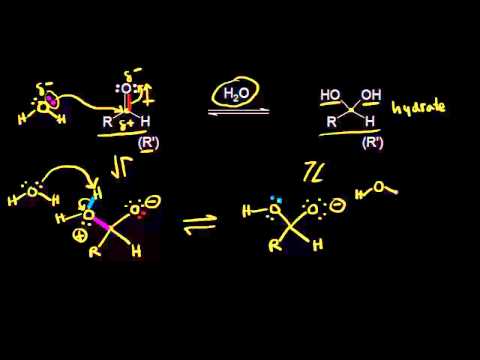
Nonetheless, several limitations need to be addressed to augment future research. This study can contribute to the literature of Khan Academy in the chemistry field. They indicated the cognitive advantage and personalized experience of using the Khan Academy. Likewise, a post-exposure self-report survey was administered to shed light on the learning experience of using the supplemental tool. Its utilization significantly increased their test scores as compared to the students who did not use it. As results revealed, its utilization as a supplemental tool had a viable impact on the students performance in chemistry. Thirty 1st year non-STEM students, equally distributed to control and experimental groups, were selected using a matched paired sampling method to participate in a two-week-long intervention. This study utilized a quasi-experimental equivalent design with a post-test only. We are waiting for the thermometer and the water to reach thermal equilibrium! At thermal equilibrium, the temperature of the thermometer bulb and the water bath will be the same, and there should be no net heat transfer from one object to the other (assuming no other loss of heat to the surroundings).This study explored the effectiveness of using the Khan academy video podcast as a supplemental tool in the chemistry class of Engineering programs. In order to make sure the reading is accurate, we usually want to wait for the temperature reading to stay constant. 
Let's say we are measuring the temperature of a water bath. Any time we use a thermometer, we are using the zeroth law of thermodynamics. 
The zeroth law allows us to measure the temperature of objects. Another way to state the zeroth law is to say that if two objects are both separately in thermal equilibrium with a third object, then they are in thermal equilibrium with each other. The zeroth law says when two objects at thermal equilibrium are in contact, there is no net heat transfer between the objects therefore, they are the same temperature. The zeroth law of thermodynamics defines thermal equilibrium within an isolated system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
